May 2, 2022
After your lightbulb moment this morning of starting a new research project on randomized controlled trials of mechanical thrombectomy in acute ischemic stroke patients, you are now settled at your desk, finalizing what type of search to run for your systematic review. You think you’ve created a pretty solid search, and it has come back with a couple hundred articles. But what do I do next?
You figure out which studies to include and exclude in your review.
Easy! Ask yourself: does this paper address the research question I am trying to answer? If it does, include it, and if it doesn’t, exclude it. That being said, it’s sometimes tricky to figure out what’s useful and what’s not for a research question, which is why we recommend that you write a protocol to explicitly define your research question, inclusion criteria, and exclusion criteria before you start screening. If you want some help on how to write a protocol in NK, take a peek at these instructions.
Once you’ve got your protocol nailed down, you can go ahead and create a list of exclusion reasons that you expect to come across. For our example, a common exclusion reason for our AIS and thrombectomy nest would be “not related to mechanical thrombectomy.” You can then exclude all studies that do not report mechanical thrombectomy under this exclusion reason while you screen.
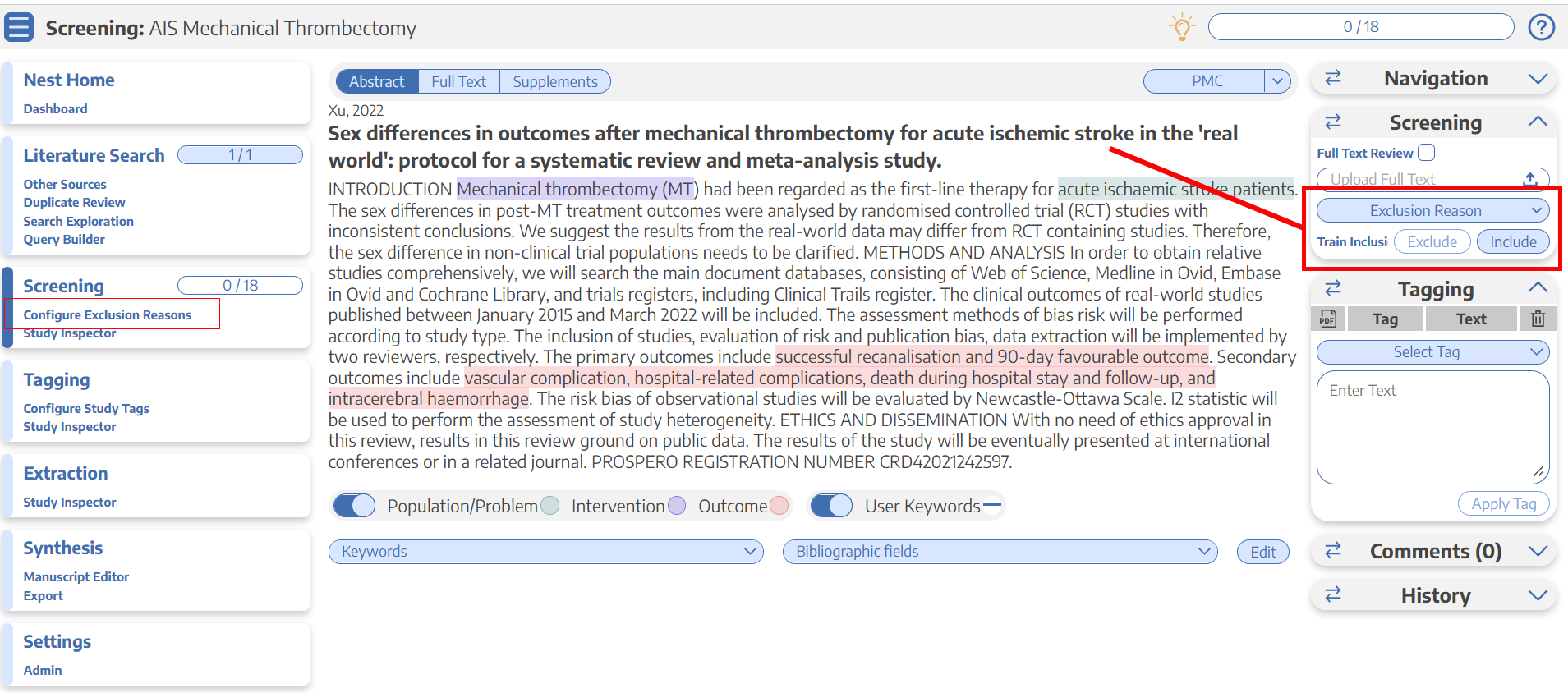
How do I include or exclude a study in the nest?
Take a peek at these instructions to learn the mechanics of it in the software. TLDR; it’s as simple as a click.
What if I need to exclude a study for a reason I didn’t put in my list?
What if I can’t screen a study because I can’t find the full text?
Advanced question: Is dual screening available?
I am so glad you asked! Yes, it is. 🙂 Check out this part of the wiki to learn how to use it.
Pro-tips:
- Upload the full texts whenever you include a study. This will save you time down the line. Trust me!
- Whatever exclusion reasons you create will be present in your PRISMA diagram. So, make sure your exclusion reasons are easy to understand.
- Whenever you upload a PDF, the software will automatically count it as a full text review instead of an abstract only review in the PRISMA diagram.
Additional resources:
Need Additional Assistance?
Email us: support@nested-knowledge.com or click the button below. We’d love to hear from you.
